Rigorous cybersecurity qualifications that stand up to regulations
Evaluate Vendor security maturity and risk with expert-led cybersecurity assessments.
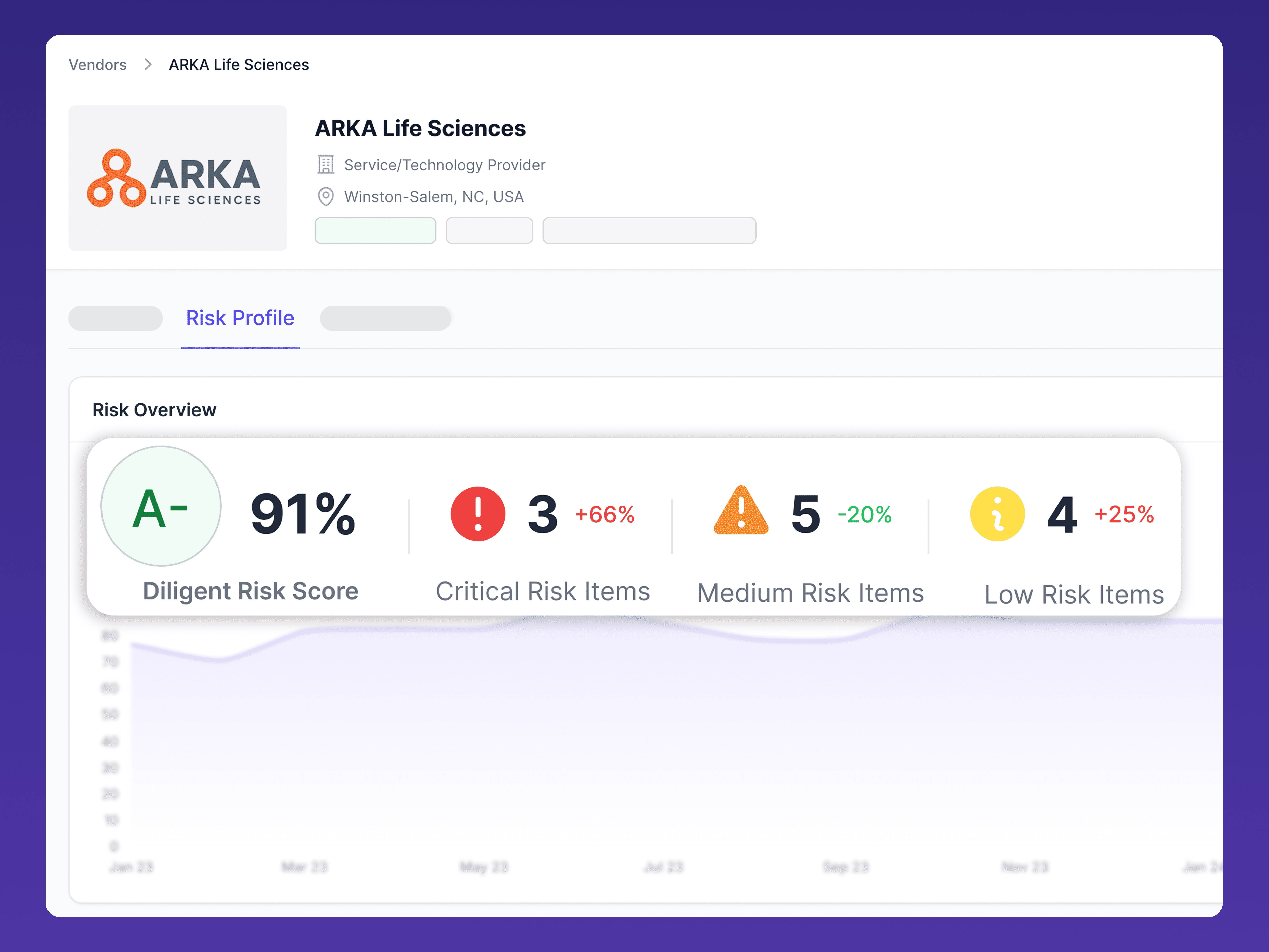
OUR FOCUS
Our expertise
Deep technical expertise for clinical environments
Our audits
Comprehensive qualification support
Thorough, industry-aligned cybersecurity assessments
A defensible, regulator-aligned assessment of a vendor’s cybersecurity posture—mapped to clinical trial requirements, quality expectations, and global privacy regulations.
Infrastructure & Systems Security
Network architecture, segmentation, and secure configuration
Cloud infrastructure posture and shared responsibility models
Server location, hosting architecture, and geographic controls
Identity & Access Management (IAM), including MFA, RBAC & least-privilege controls
Zero-trust approach and endpoint protection
Application & Platform Security
Secure development practices (SDL/SDLC)
Vulnerability management, patching cadence, and penetration testing results
API security and third-party integrations
Change management and configuration governance
Data Privacy, Protection & Governance
Data classification, encryption at rest/in transit, and key management
Data residency and jurisdictional compliance (GDPR, HIPAA, etc.)
Customer data segregation, retention, and deletion practices
Monitoring for unauthorized access or anomalous data behavior
Operational Security & Incident Preparedness
Logging, audit trails, real-time system alarms, and firewall protections
SIEM/SOC monitoring, intrusion detection/prevention systems
Incident response plans, breach notification workflows, and root-cause analysis rigor
Business continuity planning, backup frequency, and disaster recovery capabilitiesgent offers a robust database of already-completed, up-to-date GxP qualification audit reports to enable clients to qualify new Vendors in a fraction of the time and cost. Clients can also engage our team of expert auditors for new audits tailored to specific studies or Vendors
Vendor’s Vendors (Fourth-Party Risk)
Oversight and qualification of critical hosting, processing, or software dependencies
Contracts, SLAs, and security assurances from sub-processors
rESOURCES
More from Diligent
Qualify innovative Vendors with speed and rigor.
Learn how you can mitigate third-party risk and stay ahead of evolving regulations.