Independent, expert-led AI qualifications
Evaluate Vendors’ AI design, data use, and compliance with regulator-aligned assessments.
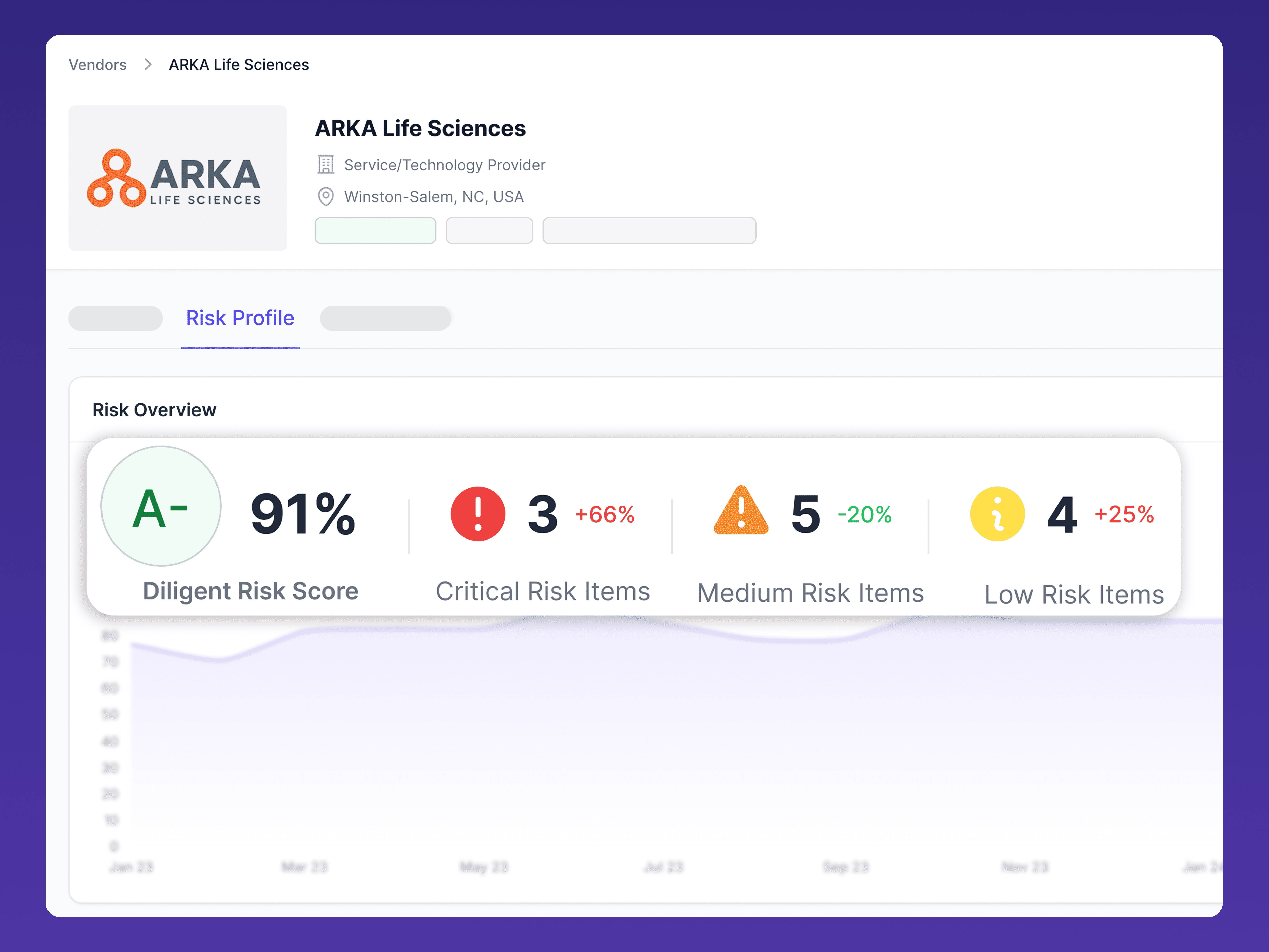
OUR FOCUS
Our expertise
Deep technical expertise for GxP environments
Our audits
Comprehensive qualification support
Thorough, industry-aligned AI assessments
Diligent evaluates the full lifecycle of a vendor’s AI capabilities, with emphasis on transparency, compliance, and ethical use.
AI Architecture & Model Design
Type of AI models used (LLMs, ML models, statistical models, generative tools)
Model provenance, training pipeline, and update cadence
Use of open-source, commercial, or proprietary models
Prompt management, model context windows, and output control mechanisms
Data Use, Training, and Privacy
Whether and how customer data is used for model training or fine-tuning
Data ingestion flows, preprocessing, and anonymization practices
Isolation of customer data from model inference/training layers
Controls to prevent unintended data retention, leakage, or memorization
Compliance with GDPR, HIPAA, and AI-specific governance frameworks
Third-Party AI & Embedded Tools
Identification of external AI services or APIs embedded in the product
Risk analysis of dependencies (e.g., model providers, ML infrastructure, vector databases)
Contractual and technical safeguards around third-party data use
AI Governance, Auditability & Safety
Documented AI policies, acceptable use frameworks, and human-in-the-loop oversight
Model monitoring, bias detection, versioning, and performance drift tracking
Explainability and transparency practices
Security and misuse prevention for model endpoints
Guardrails, red-team testing, and hallucination mitigation strategies
Regulatory Alignment & Readiness
Preparedness for evolving global AI regulations (EU AI Act, U.S. frameworks, ISO standards)
Internal expertise, accountable roles, and governance structures
SOPs, validation, and change-management processes for AI-enabled features
rESOURCES
More from Diligent
Qualify innovative Vendors with speed and rigor.
Learn how you can mitigate third-party risk and stay ahead of evolving regulations.